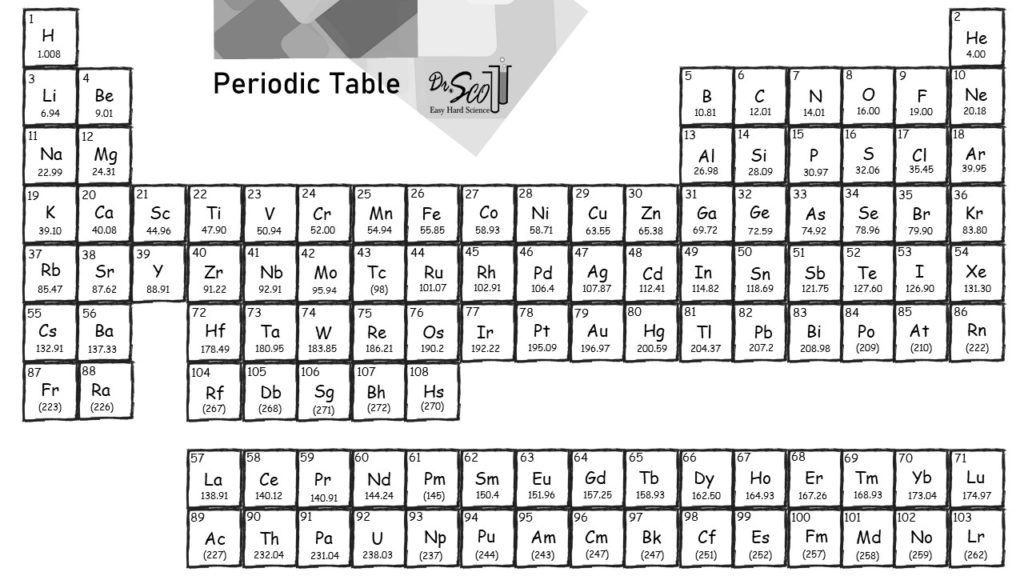
One group of elements absent from Mendeleev’s table was the noble gases, all of which were discovered more than 20 years later, between 18, by Sir William Ramsay (1852–1916 Nobel Prize in Chemistry 1904). This would mean that indium’s atomic mass was actually 113, placing the element between two other metals, cadmium and tin. Because elemental indium is a silvery-white metal, however, Mendeleev postulated that the stoichiometry of its oxide was really In 2O 3 rather than InO. If this atomic mass were correct, then indium would have to be placed in the middle of the nonmetals, between arsenic (atomic mass 75) and selenium (atomic mass 78). The atomic mass of indium had originally been reported as 75.6, based on an assumed stoichiometry of InO for its oxide. He discovered, for example, that the atomic masses previously reported for beryllium, indium, and uranium were incorrect. When the chemical properties of an element suggested that it might have been assigned the wrong place in earlier tables, Mendeleev carefully reexamined its atomic mass. Table 3.4.1 Comparison of the properties predicted by Mendeleev in 1869 for eka-aluminum and eka-silicon with the properties of gallium (discovered in 1875) and germanium (discovered in 1886) Property The observed properties of gallium and germanium matched those of eka-aluminum and eka-silicon so well that once they were discovered, Mendeleev’s periodic table rapidly gained acceptance. Two of the blanks Mendeleev had left in his original table were below aluminum and silicon, awaiting the discovery of two as-yet-unknown elements, eka-aluminum and eka-silicon (from the Sanskrit eka, meaning “one,” as in “one beyond aluminum”). 
The most convincing evidence in support of Mendeleev’s arrangement of the elements was the discovery of two previously unknown elements whose properties closely corresponded with his predictions ( Table 3.4.1 ). 
Until roughly 1960, a rectangular table developed from Mendeleev's table and based on reactivity was standard at the front of chemistry lecture halls. Notice how this approach has trouble with the transition metals. In Group VII, one atom of fluorine (F), chlorine (Cl), and bromine (Br), reacts with one atom of hydrogen. For example, in Group I, two atoms of hydrogen (H), lithium (Li), sodium (Na), and potassium (K) form compounds with one atom of oxygen. The groups in Mendeleev's table are determined by how many oxygen or hydrogen atoms are needed to form compounds with each element. Atomic masses are shown after equal signs and increase across each row from left to right. In Meyer’s plot of atomic volume versus atomic mass, the nonmetals occur on the rising portion of the graph, and metals occur at the peaks, in the valleys, and on the down slopes.įigure 3.4.3 Mendeleev’s periodic table, as published in the German journal Annalen der Chemie und Pharmacie in 1872: The column headings “Reihen” and “Gruppe” are German for “row” and “group.” Formulas indicate the type of compounds formed by each group, with “R” standing for “any element” and superscripts used where we now use subscripts. \( \newcommand\)Īs shown in Figure 3.4.2, the alkali metals have the highest molar volumes of the solid elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
